Data
Assay type
Test type
Qualitatively
Sample material
Nasopharynx | Oropharynx
Duration
10-15 minutes
Pack size
20 tests / 1 tests
Temperature
4 – 30 ° C
Durability
2 years
Sensitivity
97.6%
Specificity
98.4%
Accuracy
98.1%
€3.95/ Test
Ready for dispatch in 1-2 working days
Available from stock in Ireland
- 40 to 600 tests
€3.95
- 600 to 1200 tests
€3.70
- 1201 to 5000 tests
€3.50
- 5001 to 10,000 tests
€3.30
- Over 10,000 tests
From €2.95 On demand
- Sale only to authorized groups of customers within the meaning of §3 MPAV
- No sale to private individuals
- Use only by trained medical professionals
- 20 tests in the kit
- Minimum purchase 40 tests
- Available from stock in Ireland

Test Principle
Product Variants

Efficiency at Optimum Cost
This package is mainly targeted at large clients, such as doctors, nursing homes and high-performing clinics. Twenty quick samples, swabs and a large buffer bottle constitute the test package.

Optimum Flexibility
This kit is mainly targeted at customers who do not want to carry out all the tests at the same time or who want to work more easily. There are 20 rapid samples, swabs and 20 sample tubes containing the sample buffer included in the package.

Outer Individual Packaging
This item is specifically targeted at resellers or individual consumers. Every test kit consists of a single rapid test, a swab and a pre-filled buffer sample tube.
Documents

Quality control
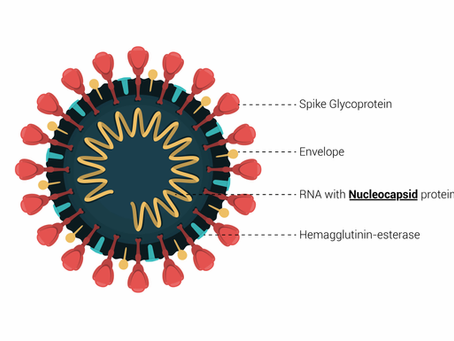
infection successfully with Lateral Flow Assays
As a manufacturer of in-vitro diagnostics, seamless quality assurance is a matter of course for us. In addition, all processes that contribute to development and production are certified according to ISO13485 by TÜV Süd. Furthermore, all management processes are certified according to ISO9001 in order to offer the best possible quality. All products are CE compliant and are sold worldwide.
Please contact us if you want to find out more about the quality assurance of our products.