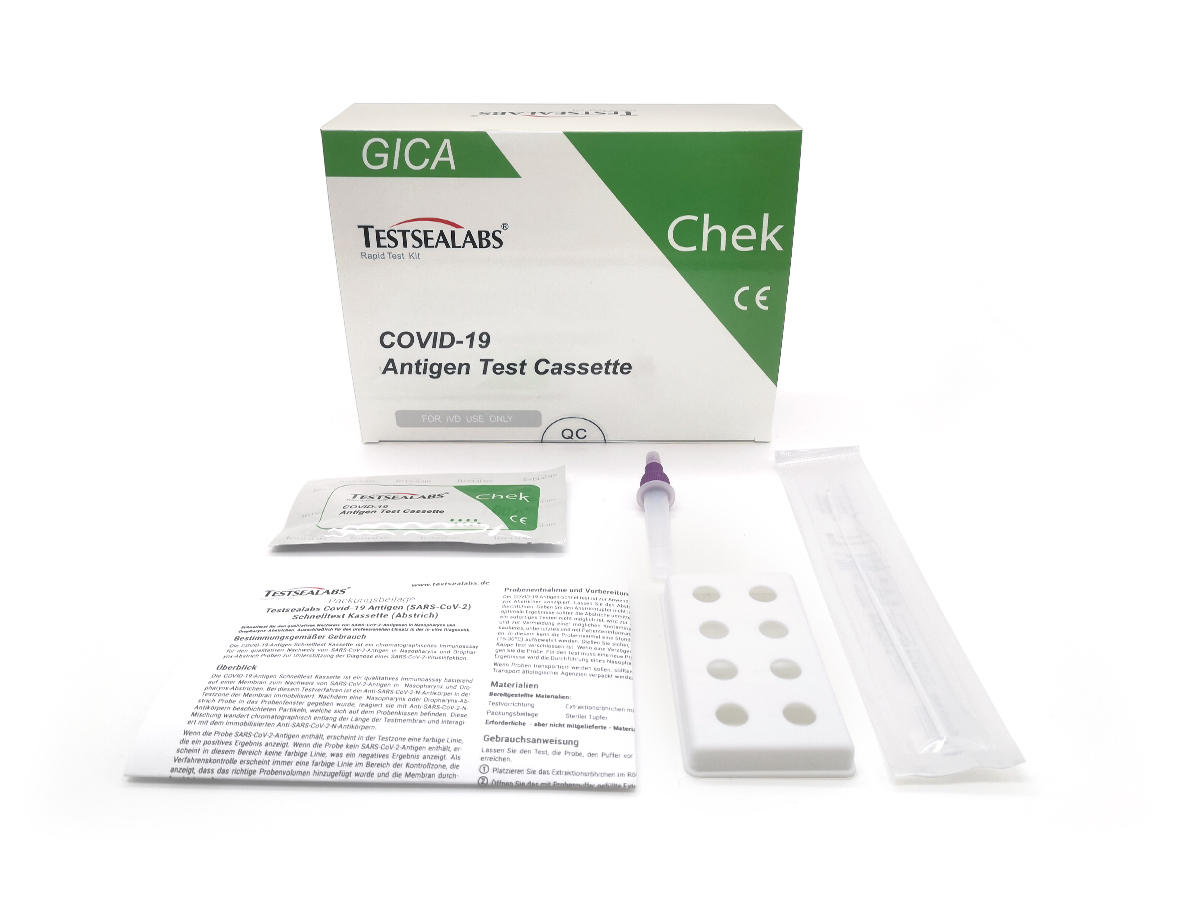
Testsealabs® Antigen rapid test for direct detection of the pathogen of the coronavirus SARS-CoV-2
- Sale only to authorized groups of customers in the sense of §3 MPAV
No sale to private individuals - Use only by medically
trained personnel - 20 tests in the kit
- Minimum purchase 40 tests
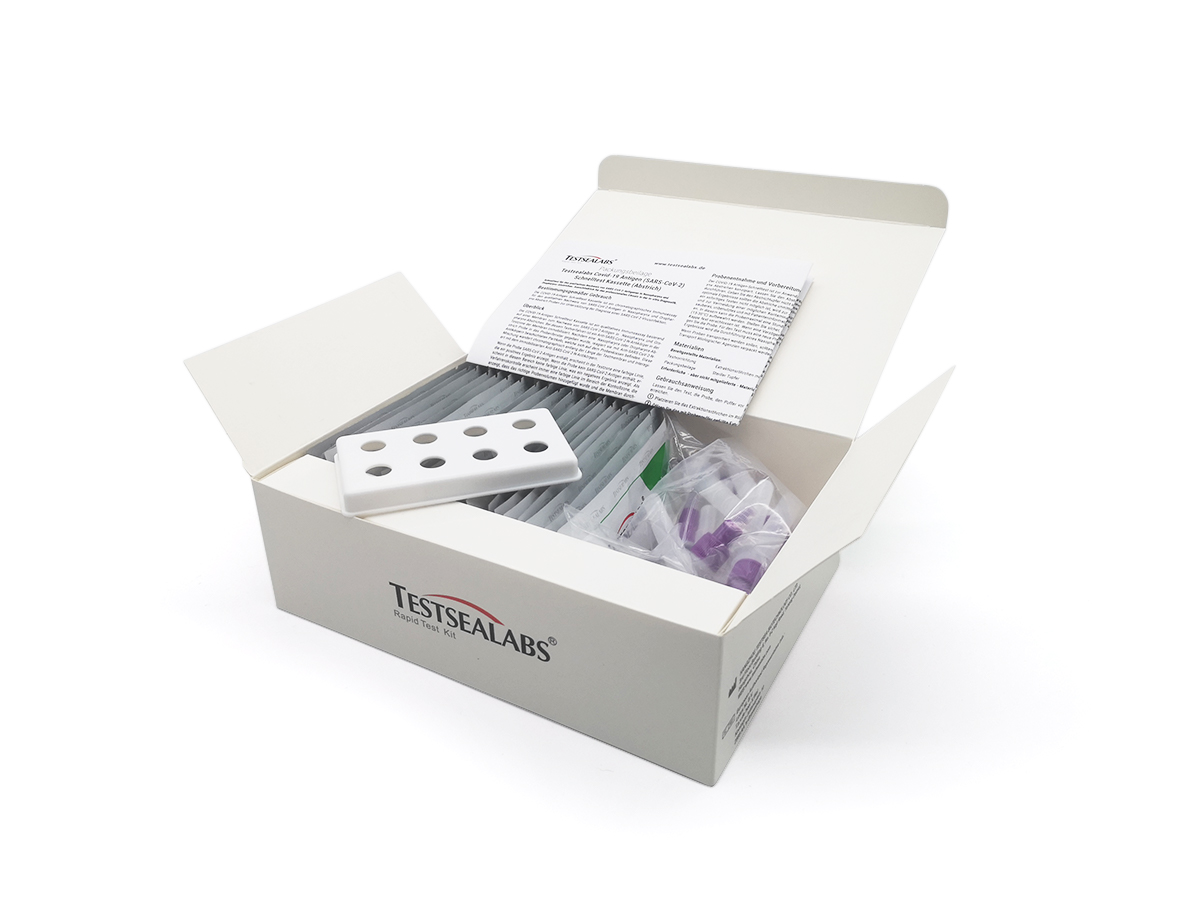
scope of delivery
- 1x package insert
- 1x tube stand
- 20 x test cassette
- 20x sterile swabs
- 20x collection tubes with cap and pre-filled test liquid
Shipping within two days. Delivery time for large quantities on request.
This kit is aimed primarily at users who do not want to carry out all tests at the same time or who want particularly user-friendly and quick handling. One sales unit contains 20 rapid tests, swabs and 20 sample tubes, which already contain the sample buffer.
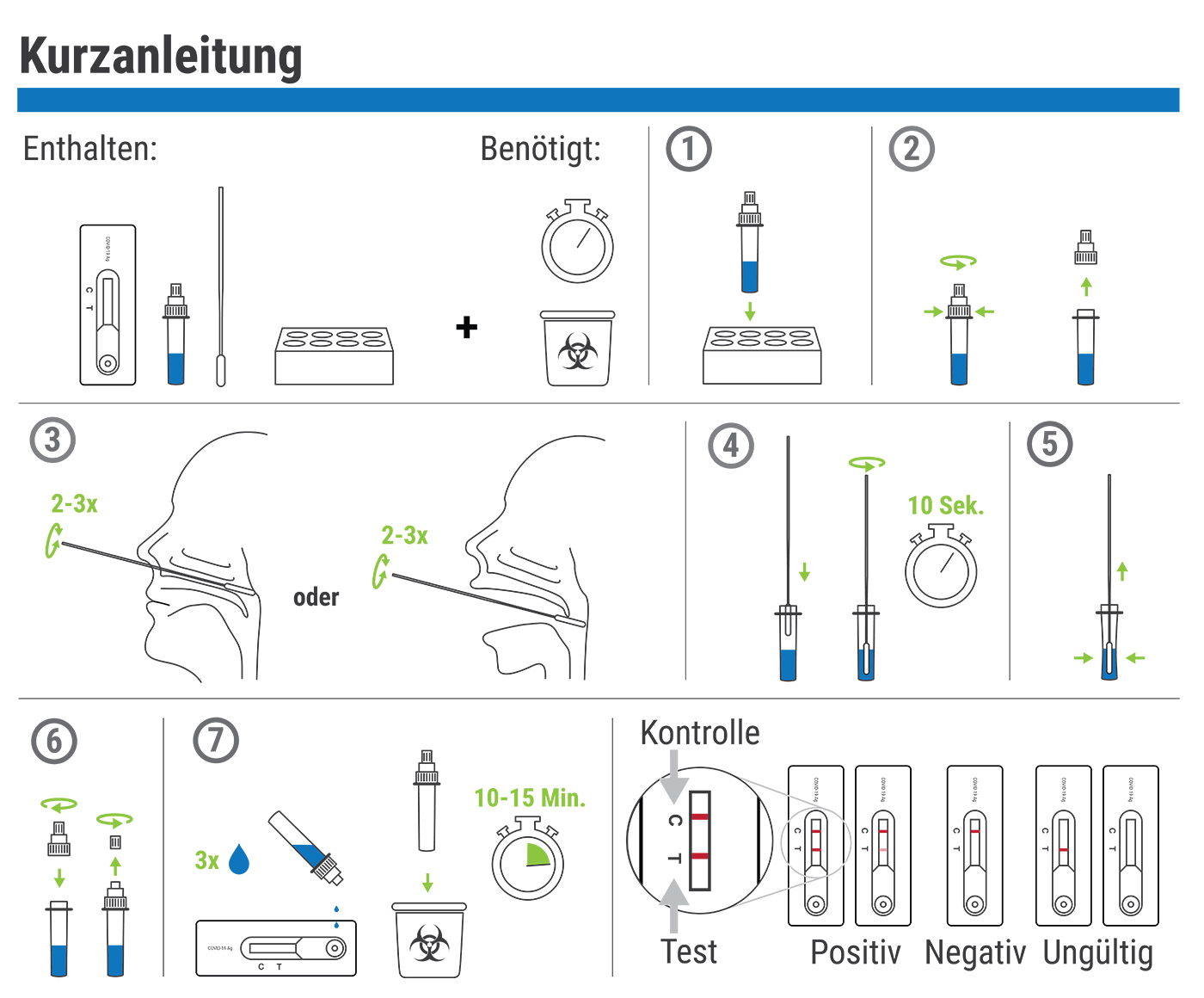
The Testsealabs COVID-19 antigen rapid test can be carried out on site by trained staff and directly detects the causative agent of COVID-19 and its antigens. To do this, a smear is taken from the back of the nose or throat and applied to a test cassette. This works according to the lateral flow principle and shows within 10 to 15 minutes whether COVID-19 antigens could be detected in the sample.
- CE certified and reimbursable test listed at the Federal Institute for Drugs and Medical Devices according to the Coronavirus Test Ordinance (TestV)
under the number AT 082/20 . - Carried out on site by medically trained staff,
results after 10 to 15 minutes - Reliable exclusion of a Sars-CoV-2 infection with high accuracy
- Can be performed as a nasopharynx or oropharynx smear
- The test is developed as a Point of Care (POC). No equipment is required, all consumables are included
Performance data
The performance data of the test meet the requirements of the Robert Koch Institute (RKI) for rapid antigen tests as part of the nationwide test strategy. The performance data refer to the current PCR gold standard.
Sensitivity: 97.6%, 95% CI (94.9% – 100%)
Specificity: 98.4%, 95% CI (96.9% – 99.9%)
Accuracy: 98.1%
Product overview: Product overview PDF
Package insert: Package insert PDF
Further product data can be found at www.testsealabs.ie
Note: Currently not available shipping to Australia.